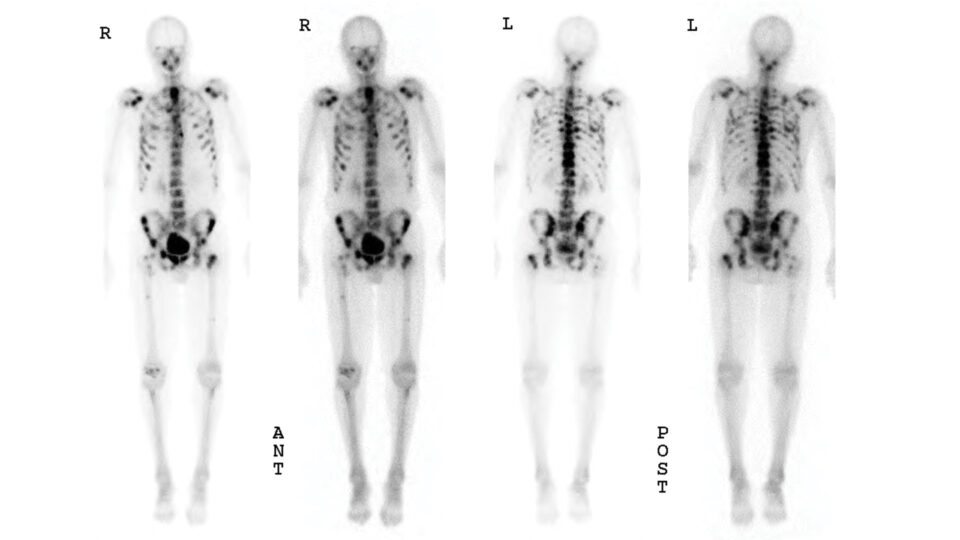
Solid tumors have been a formidable target for cellular therapies. Unlike hematologic malignancies, solid tumor cells are embedded within hard-to-reach tumor microenvironments that are immunosuppressive and hostile to treatments. Even when cellular therapies reach the tumor and overcome these immunosuppressive defenses to engage cancer cells, there are several remaining hurdles, including ensuring that treatments retain antitumor specificity, that they are able to access heterogenous and/or intracellular antigens, and that they do not cause excessive side effects.
Medical oncologist Salman R. Punekar, MD, is leading multiple clinical trials investigating novel approaches to overcoming these challenges, including logic-gated CAR T cells and armored T cell receptor (TCR) therapy. He and colleagues at Perlmutter Cancer Center are applying these strategies and others to go after prominent targets like KRAS, EGFR, mesothelin, CEA, and DLL3.
Here, Dr. Punekar discusses these emerging cell therapy technologies, highlights their early success, and looks ahead to opportunities to expand access—including with in vivo approaches.
Physician Focus: One next-generation cellular therapy that NYU Langone is helping pioneer is logic-gated CAR T cells. Can you tell us how it works?
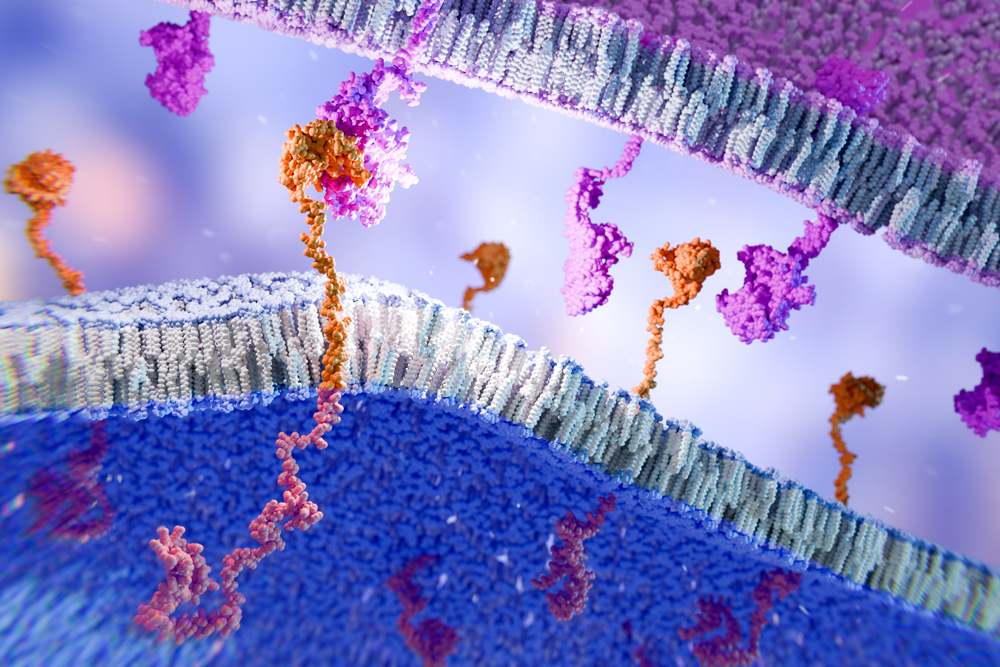
Dr. Punekar: Logic-gated CAR T cells come in a variety of flavors, and usually are “AND”, “OR” or “NOT” logic gates. In this case, we are discussing NOT gates, in which the engineered T cell has two CARs: one is an activating receptor and one is a blocking receptor. If you’re targeting EGFR, for example, and the CAR T cell engages EGFR on a normal cell, it will get an activation signal. But it will also engage human leukocyte antigen (HLA), which is found on all normal cells, and that blocks the CAR T cell activation.
In tumor cells that lose HLA in what’s called HLA loss of heterozygosity, the CAR T cell again engages and activates because it recognizes EGFR. But since there’s no HLA, there’s nothing to turn on the blocker of the activation, and so the CAR T cell kills the tumor cell.
“We recently reported a stage 4 non-small cell lung cancer patient with KRAS and STK11 co-mutations who achieved a complete response after mesothelin logic-gated CAR T cell therapy.”
Salman Punekar, MD
NYU Langone has led that logic-gated technology across multiple targets, including CEA, mesothelin, and EGFR. In fact, we recently reported a stage 4 non-small cell lung cancer patient with KRAS and STK11 co-mutations—one of the worst types of lung cancer—who achieved a complete response after mesothelin logic-gated CAR T cell therapy. It’s the first time that a CAR T cell therapy has resulted in a complete response for a patient with lung cancer.
Physician Focus: What’s the next step in refining that therapeutic approach?
Dr. Punekar: That case was part of the phase 1/2 EVEREST-2 trial and used an earlier generation CAR T cell. We’re now running a new arm of the trial to test a next generation “armored” version. It has a membrane-tethered IL-12 cytokine. When it reaches its target, IL-12 gets presented locally and induces more proliferation and activation of the T cells.
Physician Focus: Can you tell us about your ongoing efforts to test and improve TCR therapies directed against KRAS-mutated cancers?
Dr. Punekar: The majority of KRAS treatments focus on small molecules. This is an alternative, complementary way to use a cell therapy within the KRAS treatment space, and it’s part of NYU Langone’s newly designated KRAS Excellence Initiative.
In a phase 1 dose escalation study in patients with locally advanced or metastatic solid tumors harboring KRAS mutations, we’re evaluating a next-generation TCR with a TGF-beta knockout, which is basically an armoring technique to increase its effectiveness against solid tumors. This trial was the impetus for a larger collaboration between AstraZeneca and NYU Langone, which is paving the way for other new cellular therapy trials.
Physician Focus: DLL3 targeting has emerged as a potentially ground-breaking strategy for small cell lung cancer. How are you approaching cell therapy here?
Dr. Punekar: There’s a very interesting trial for recurrent small cell lung cancer that we’ve been collaborating on with a biotech company.
Small cell lung cancer has been revolutionized by DLL3-targeting therapy, most notably with the approval of an immunotherapy drug called tarlatamab, which is a T cell engager that has shown promise in linking T cells to DLL3-expressing tumors. We hope that new CAR T cells targeting DLL3 will further change the treatment landscape for small cell lung cancer.
In this case, NYU Langone physicians collaborated early on to help develop the strategy for this trial and will be sharing in the science as a true partnership between academia and biotech.
“One of the biggest things that we’d like to figure out is how we can make cell therapy accessible to everyone.”
Physician Focus: What’s next on your to-do list for improving cellular therapy?
Dr. Punekar: A lot of these trials are very restrictive on who can enter, meaning you must have a certain HLA type and a certain mutation or biomarker. One of the biggest things that we’d like to figure out is how we can make cell therapy accessible to everyone. We would like to see many more HLA types represented as well as treatments for many more mutations and biomarkers.
The second thing we’re working on is further improving the armoring techniques so that we can make the T cells more potent. And the third thing would be to reduce the risk of on-target, off-tumor toxicity. As an example, if you have something that’s directed at mesothelin, we need it to kill only mesothelin-expressing cancer cells and not mesothelin-expressing lung or gastrointestinal cells. There are a variety of techniques that we utilize to do that, including the logic-gated technology.
The fourth thing we need to figure out is the logistics of CAR T cells and TCRs. They cost a lot of time and money and sometimes require apheresis. We’re thinking about newer technologies such as in vivo CAR T cell therapy, which would enable the production of CAR T cells within the patient, so you’re doing the manufacturing in real time. It costs much less and permits larger-scale trials. There’s a lot of opportunity there.
Disclosures
Dr. Punekar serves on the Clinical Advisory Board of A2Bio and on the SRC Advisory Board for Moonlight Bio. He is the principal investigator for cellular trials sponsored by AstraZeneca, A2Bio, Moonlight Bio, and Iovance.