Surgical management of giant hydronephrosis presents unique challenges. While nephrectomy via an open approach is feasible, patients can benefit from a minimally invasive, robotic approach to minimize perioperative morbidity and recovery time. Controlled drainage of the hydronephrotic kidney is critical to establish laparoscopic access. Advancements in robotic instrumentation have improved the platform’s capability for complex retraction and exposure. These are critical aspects of these and other cases wherein anatomic landmarks are distorted and/or unreliable.
In this Case of the Month, we present a young male with incidentally discovered giant right hydronephrosis and contralateral ureteropelvic junction (UPJ) obstruction, which required a robotic-assisted right nephrectomy and left pyeloplasty. We highlight the role of specialized retractors, flexible ureteroscopy with near-infrared fluorescence, and surgical adaptations such as judicious use of non-transecting techniques to enhance safety and optimize outcomes.
Case Highlights:
- A male in his 30s was incidentally found to have massive right hydronephrosis and mild left hydronephrosis and elected for robotic surgery.
- The right kidney was partially drained, with 2.5 L removed, and a small grasping retractor facilitated nephrectomy.
- A left UPJ obstruction was confirmed, prompting placement of a ureteral stent and scheduling of pyeloplasty six weeks after nephrectomy. The ureteral stent was removed one week prior to surgery to allow “ureteral rest” and precise assessment of the stricture.
- Left pyeloplasty with intraoperative ureteroscopy was performed using a non-transecting ureteropelvic anastomosis to preserve blood supply to the repair.
Patient Case
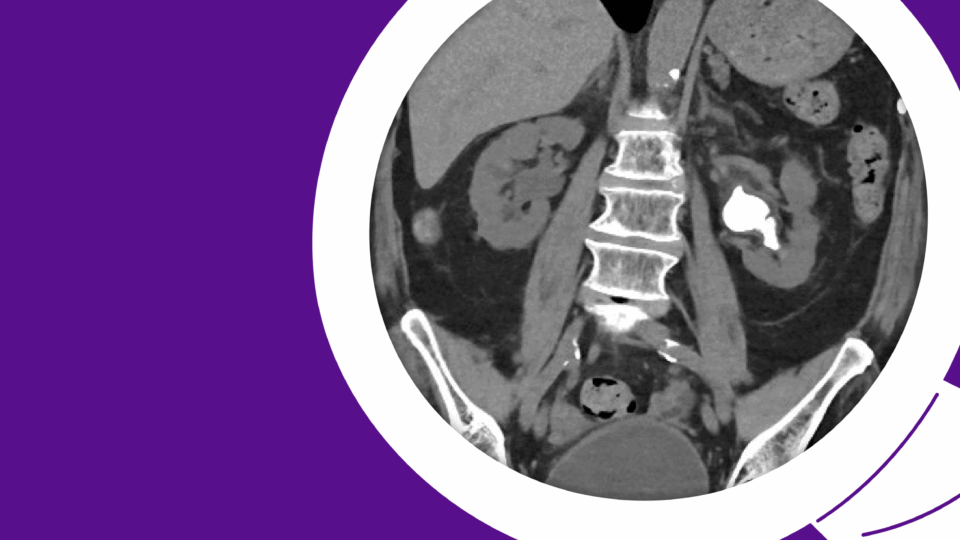
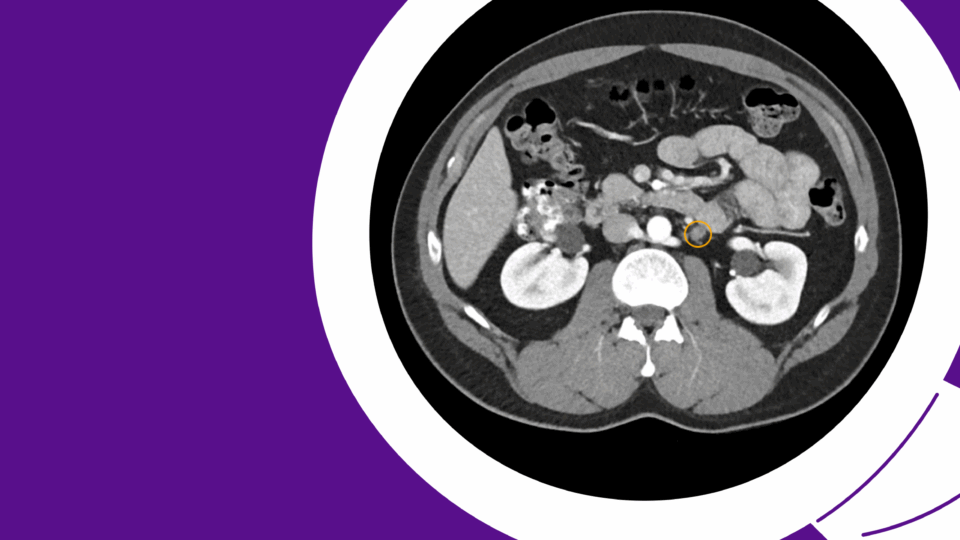
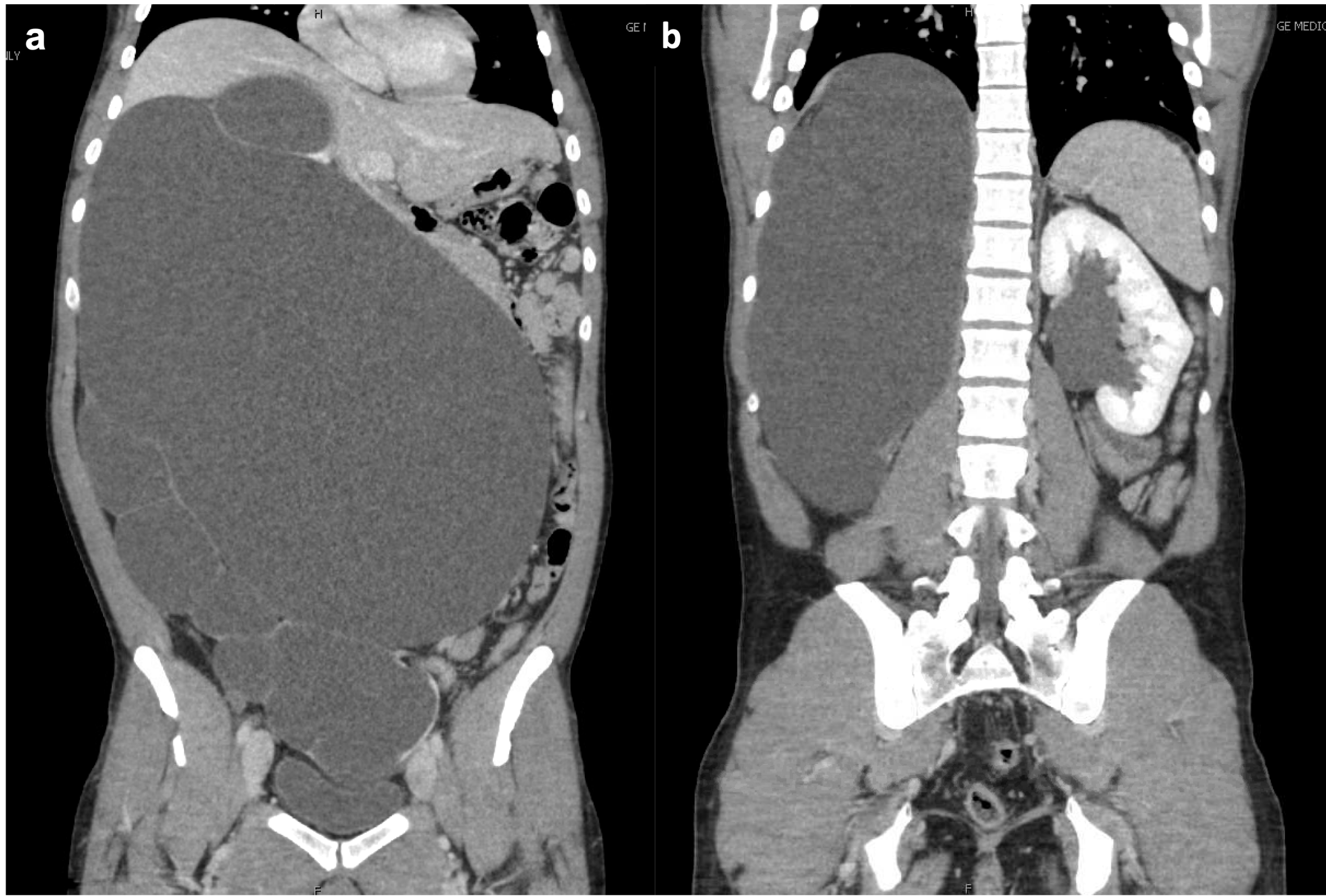
A healthy male in his early 30s had an incidental finding on abdominal sonography of a large, fluid-filled mass occupying the majority of his abdomen. Subsequent CT imaging showed massive right hydronephrosis with loss of renal parenchyma and mass effect on the intra-abdominal contents (Figure 1a). Mild left (contralateral) hydronephrosis was also noted (Figure 1b). He was subsequently referred to our institution for management.
On initial evaluation, the patient denied abdominal discomfort, flank pain, early satiety, or dyspnea. Bloodwork showed normal renal function (creatinine 1.1 mg/dL, estimated glomerular filtration rate [eGFR] 93 mL/min/1.73 m2). The patient had always presumed he had a “beer belly”, though, as noted by his wife, he does not consume alcohol. He had no known history of antenatal hydronephrosis, growth restriction, or renal insufficiency. Examination revealed a distended abdomen without tympany or tenderness.
Preoperatively, a Lasix renogram was obtained to evaluate the left-sided hydronephrosis. This was suggestive of a nonfunctional right kidney and a partial left UPJ obstruction. After discussion, the patient opted to proceed with robotic-assisted right nephrectomy and simultaneous fluoroscopic evaluation of the left collecting system.
Management
Access. Initial laparoscopic access was facilitated by percutaneously draining the fluid-filled right kidney under ultrasound guidance. A total of 2.5 L of straw-colored fluid—presumably urine—was evacuated, and serial examination showed progressive improvement of abdominal distension. Once the fluid was partially evacuated, a Hasson cut-down was performed to place the initial laparoscopic assistant port and insufflate the peritoneal cavity. The remaining robotic ports were placed under direct vision.
Nephrectomy. A robotic right nephrectomy was performed in the standard fashion. The renal hilum was aberrant in origin and controlled with a series of locking clips. The right ureter was transected at the UPJ and on visual inspection appeared stenotic.
At the conclusion of the procedure, the patient was repositioned and a retrograde pyelogram performed on the contralateral side. This demonstrated a left UPJ obstruction (Figure 2).
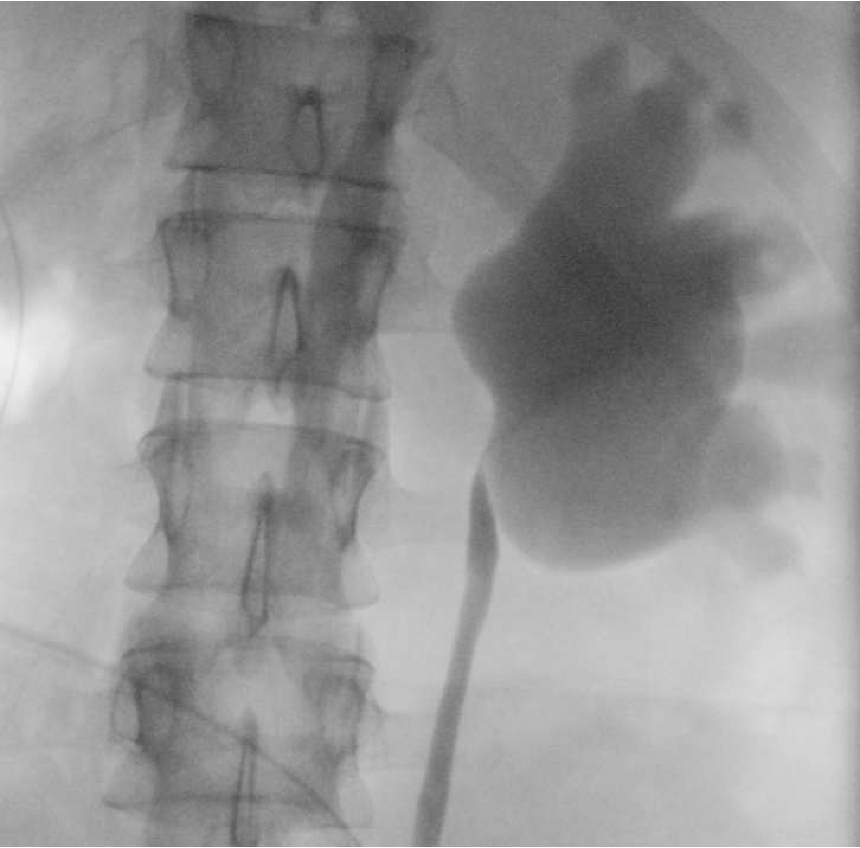
After discussion with his family, we opted to return for reconstruction of the left collecting system at a later date. A ureteral stent was placed to decompress the system and protect his functionally (and now anatomically) solitary kidney in the interim. He recovered without incident and creatinine subsequently nadired at 0.87 mg/dL (eGFR; 118 mL/min/1.73 m2).
Pyeloplasty. Six weeks following nephrectomy, the patient returned for a robotic-assisted left pyeloplasty. The left ureteral stent was removed one week prior to allow for a period of ureteral rest, and an interval basic metabolic panel was obtained to rule out acute kidney injury.
Intraoperatively, flexible ureteroscopy was performed to rapidly identify the left proximal ureter under near-infrared (Firefly) fluorescence, as well as to confirm luminal narrowing at the UPJ (Figure 3). There was no crossing vessel.
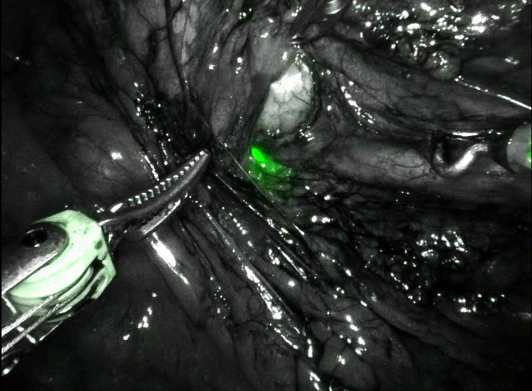
A new, dependent UPJ was fashioned over a ureteral stent as a non-transecting anastomosis (Figure 4). Intravenous indocyanine green (ICG) was given to confirm satisfactory perfusion to the repair prior to completion.
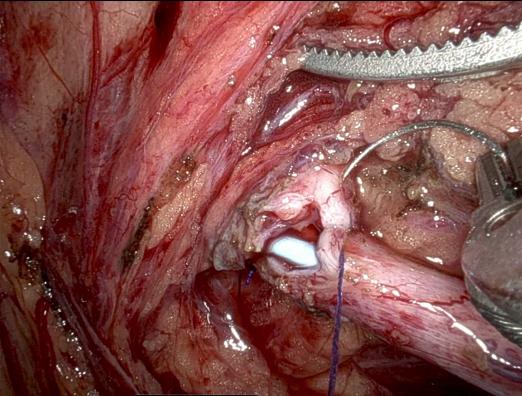
Follow-Up. The patient recovered well from his second surgery and the ureteral stent was removed after six weeks. He remains asymptomatic and will undergo periodic surveillance imaging. His most recent serum creatinine is 0.87 mg/dL (eGFR; 118 mL/min/1.73 m2).
Discussion
Antenatal hydronephrosis is a common congenital urologic abnormality and associated with 1 to 5 percent of pregnancies. Of these, a reported 7.7 percent are ultimately diagnosed with UPJ obstruction.1 An estimated 10 to 40 percent of cases are bilateral.2,3 While initial conservative management may be appropriate, surgical intervention is indicated in cases of symptomatic obstruction, recurrent infections, and renal function deterioration.4,5 In the case presented herein, we suspect the patient had a longstanding, asymptomatic right UPJ obstruction and partial left UPJ obstruction.
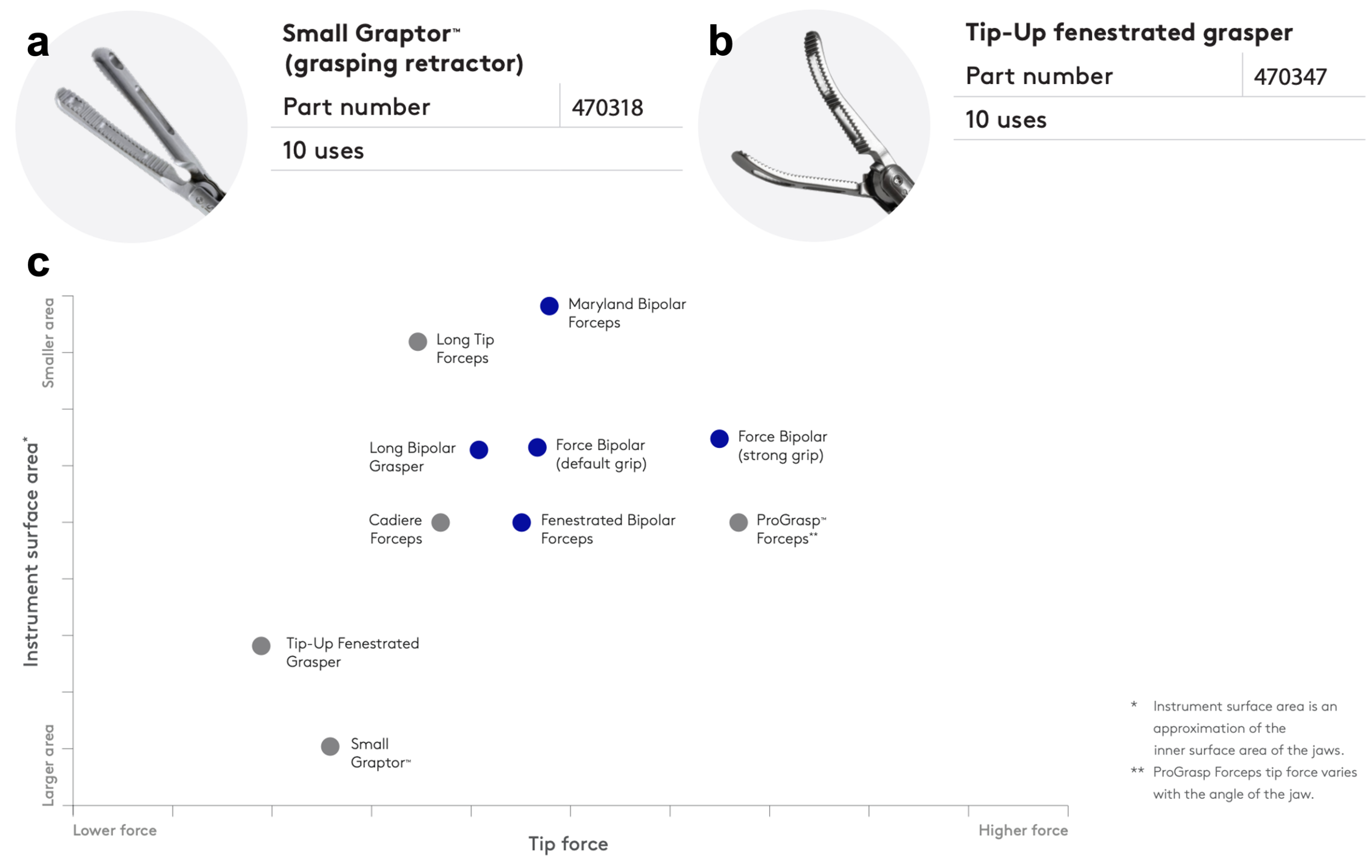
Nephrectomy. For atypical cases such as the massively hydronephrotic kidney, certain adjustments facilitate laparoscopic access and dissection. Controlled drainage of the hydronephrotic kidney is paramount; inadequate emptying can complicate initial access and port placement, whereas over-evacuation yields a floppy, voluminous sac which is difficult to handle. Inadvertent entries during the course of dissection can be managed with surgical clips or suture closure. For such cases, the da Vinci Small Graptor™ grasping retractor (Figure 5a) features a large profile well-suited to broad retraction and exposure. An alternative is the Tip-Up fenestrated grasper (Figure 5b); this has weaker grip strength, which can impede extirpative procedures but is useful for handling bowel (Figure 5c).
Pyeloplasty. For patients with ureteral strictures, we typically remove indwelling ureteral stents at least one week prior to reconstruction. This practice of “ureteral rest”, adapted from similar practices for urethral stricture disease, allows the exact location, length, and caliber of the stricture to be delineated prior to repair.6 Some authors have reported superior patency rates associated with ureteral rest, though data are mixed.7,8,9 There is no consensus as to the exact duration of “rest” necessary.
Intraoperative ureteroscopy is a helpful adjunct when a qualified assistant is available. In appropriate settings, this allows for rapid ureteral identification and stricture localization. Moreover, this practice preserves ICG for intravenous use to ensure satisfactory vascularity to the subsequent anastomosis.10 This is particularly salient for cases with impaired ureteral perfusion or those involving bowel harvest, as seen with re-operative procedures, abdominopelvic radiation, and urinary diversions. In our experience, visualizing the ureteroscope under near-infrared fluorescence can be difficult if the surrounding fibrosis is severe. Additional maneuvers include distending the affected ureter via an indwelling nephrostomy tube or pre-placed ureteral catheter, as well as manipulating a wire within the ureteral lumen while assessing for transmitted movement within the retroperitoneum.
While the Anderson-Hynes dismembered pyeloplasty is appropriate for UPJ obstruction due to crossing vessels, non-transecting anastomotic techniques have gained favor.11 In select patients without secondary intraluminal narrowing, a vascular hitch may be a viable option even in the presence of a crossing vessel.12 For recurrent UPJ obstruction, additional reconstructive options include ureteral augmentation with a buccal mucosa graft and ureterocalicostomy.13,14
References
- Fwu CW, et al. Urology. 2024;183:185-191. DOI.
- Lee YS, et al. Urology. 2013;81(4):873-878. DOI.
- Karnak I, et al. Pediatr Surg Int. 2008;24(4):395-402. DOI.
- Tabari AK, et al. J Pediatr Surg. 2020;55(9):1936-1940. DOI.
- Sadghian M, et al. Pediatr Surg Int. 2023;39(1):147. DOI.
- Terlecki RP, et al. Urology. 2011;77(6):1477-1481. DOI.
- Lee Z, et al. Urology. 2021;152:160-166. DOI.
- Campbell K, et al. J Endourol. 2026;40(2):229-232. DOI.
- Zhou Y, et al. World J Urol. 2025;43(1):186. DOI.
- van der Poel HG, et al. Eur Urol Focus. 2018;4(5):665-668. DOI.
- Amón Sesmero JH, et al. J Endourol. 2016;30(7):778-782. DOI.
- Kim JK, et al. BJU Int. 2022;129(6):679-687. DOI.
- Chao BW, et al. Urology. 2025;197:174-179. DOI.
- Xie B, et al. Int Urol Nephrol. 2024;56(6):1899-1909. DOI.