Seminal vesicle (SV) recurrence is technically challenging to treat. Salvage SV ablation alone is complex and limited by the risk of thermal injury to adjacent structures, while surgical SV excision alone may leave residual disease at the prostate–SV junction.
To address this challenge, our group at NYU Langone developed a combined salvage approach that incorporates focal cryoablation and robotic seminal vesiculectomy. In this strategy, cryoablation targets residual intraprostatic disease and the prostate–SV junction, while robotic excision enables definitive removal of involved SV tissue.
Here, we demonstrate the feasibility and tolerability of this combined approach in a heavily pre-treated patient. At one year follow-up, there is no radiographic evidence of recurrence.
This case highlights two key points. First, as recognition of SV recurrence increases, this combined technique warrants broader consideration in the management of radiorecurrent prostate cancer with SV involvement. Second, although preoperative imaging suggested disease confined to the right SV, final pathology revealed contralateral involvement of the left SV. This finding reinforces observations from our group and others that contemporary imaging may fail to detect microscopic contralateral SV disease.
Case Highlights:
- The patient’s initial intermediate-risk disease at the right base and first recurrence were treated with IMRT, ADT, pelvic nodal radiation, and HDR brachytherapy.
- PSMA PET/CT, mpMRI, and biopsy confirmed second recurrence involving the right posterior mid-gland, right SV, and right vans deferens.
- Combined salvage cryoablation and robotic seminal vesiculectomy was performed, with retention of a thermocouple at the cryoablation margin serving as a landmark to guide the proximal extent of robotic resection.
- Excised SV specimens lacked radiation treatment effect despite prior radiation, highlighting their potential as sites of recurrence.
Case Presentation
Initial Diagnosis and Treatment Prior to Referral to NYU Langone (2014). A male in his early 70s with coronary artery disease, atrial fibrillation (on dabigatran and digoxin), and hyperlipidemia was diagnosed in 2014 with favorable intermediate-risk prostate cancer (PSA 4.1 ng/mL). Initial biopsy showed Grade Group 2 disease in the right lateral base and multifocal Grade Group 1 disease elsewhere. He underwent definitive intensity-modulated radiation therapy (IMRT) (81 Gy in 45 fractions), completed in 2015, with PSA nadir of 0.9 ng/mL at 18 months. Pelvic nodes were not treated initially.
First Recurrence and Salvage Treatment (2019–2022). Biochemical recurrence was identified in 2019 (PSA 4.1 ng/mL). Axumin PET/CT and MRI suggested possible nodal recurrence, prompting androgen deprivation therapy (ADT) (October 2020–August 2021), followed by salvage pelvic nodal radiation (56.25 Gy in 25 fractions).
PSA declined to 0.05 ng/mL but rose again to 0.91 ng/mL by August 2022. MRI showed no local recurrence; however, Ga-68 prostate-specific membrane antigen (PSMA) PET demonstrated focal uptake in the prostate without nodal or distant disease. Biopsy confirmed recurrent Grade Group 3 disease in the right lateral base and Grade Group 2 disease in the right base.
He then underwent salvage high dose rate (HDR) brachytherapy boost (11 Gy × 2 fractions), completed in December 2022, achieving a PSA nadir of 0.34 ng/mL.
Referral to NYU Langone and Identification of Second Recurrence (2024). By October 2024, PSA had risen to 1.23 ng/mL with testosterone 590 ng/dL, confirming non-castrate recurrence. He had mild lower urinary tract symptoms controlled on tamsulosin and baseline erectile dysfunction.
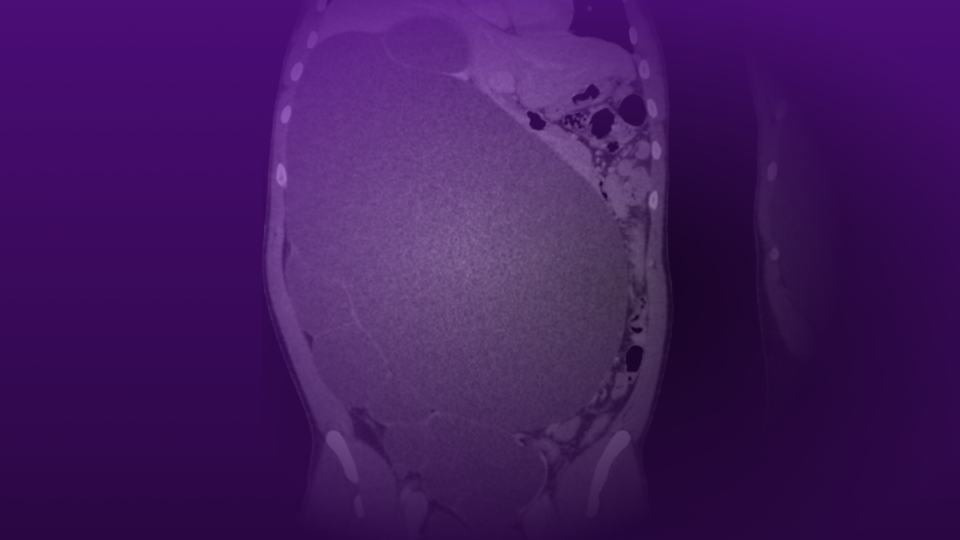
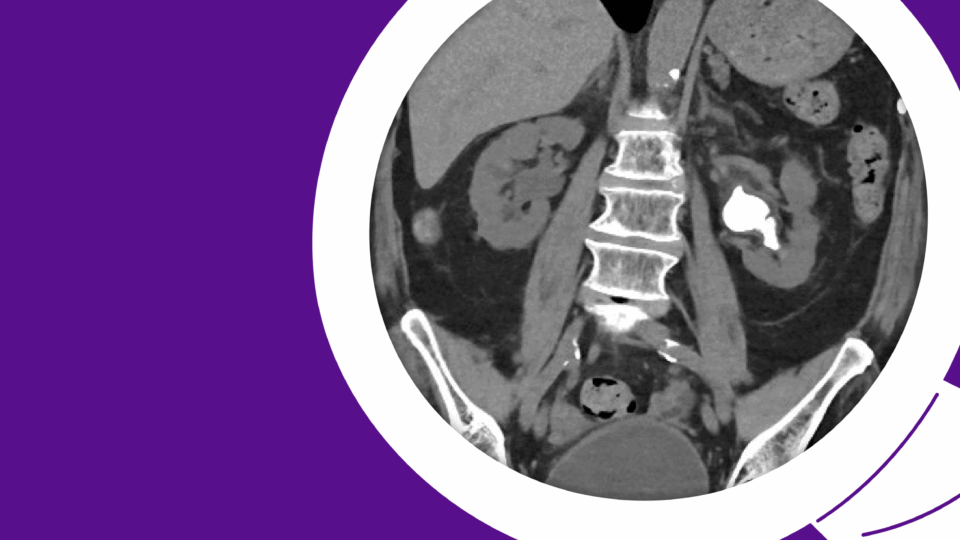
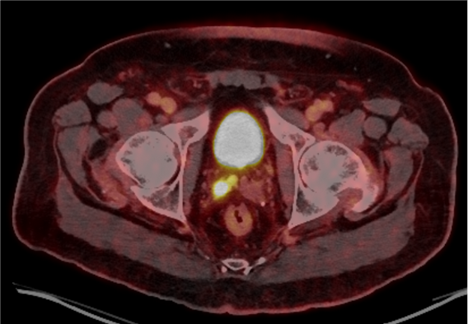
PSMA PET/CT demonstrated persistent uptake in the right posterior mid-gland and new uptake in the right SV, without nodal or distant disease (Figure 1). Multiparametric MRI (mpMRI) confirmed recurrent tumor involving the right SV and right vas deferens (Figure 2).
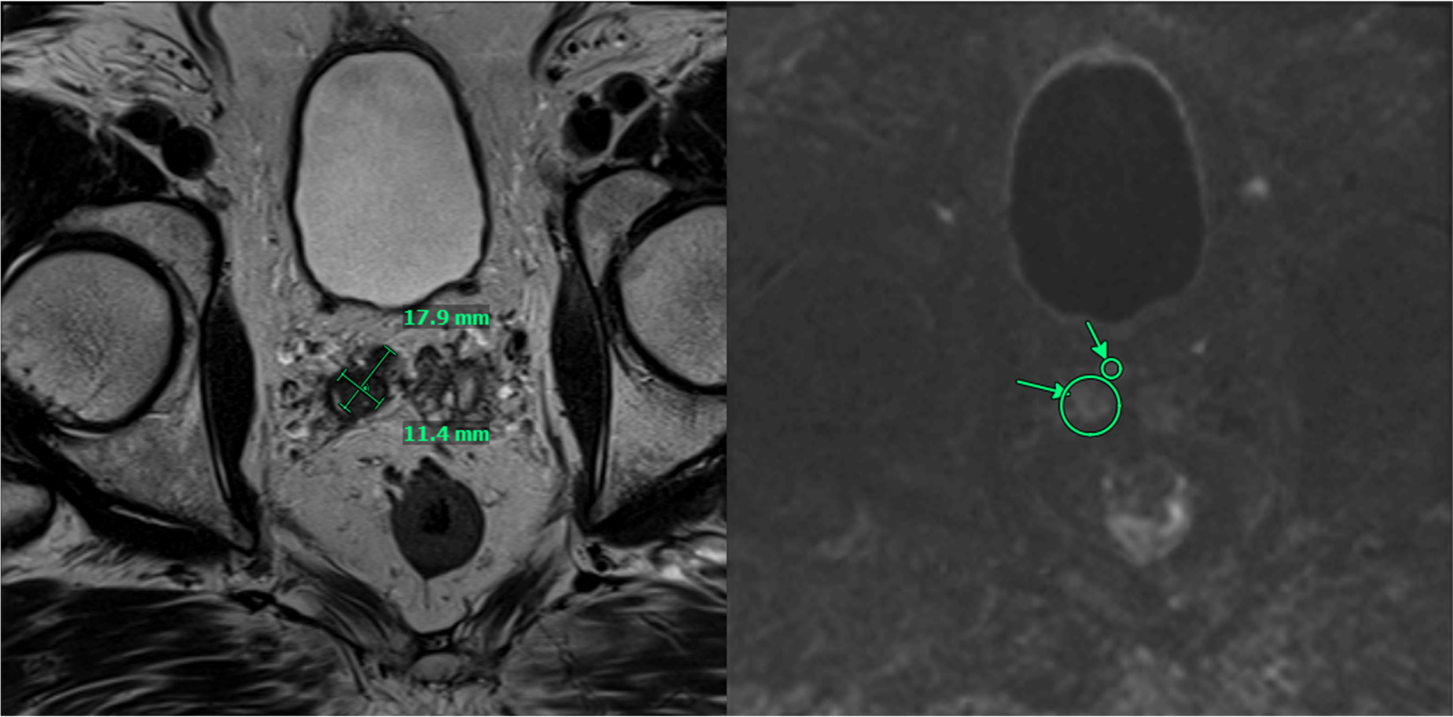
January 2025 transperineal biopsy showed no cancer in 13 systematic cores; targeted biopsy of the right SV demonstrated Grade Group 2 adenocarcinoma in 1/4 cores (up to 2.5 mm). PSA at biopsy was 1.35 ng/mL.
After multidisciplinary counseling, he elected salvage focal prostate cryoablation with robotic seminal vesiculectomy, performed in March 2025.
Management
Combined Salvage Cryoablation and Robotic Seminal Vesiculectomy. Cryoablation was performed first using two treatment probes and three thermocouples placed under ultrasound guidance to treat the right prostate base–SV junction. Two freeze-thaw cycles were completed, achieving nadir temperatures of −83°C and −79°C at the base/SV junction target and −42°C to −43°C at posterior and right posterolateral treatment margins.
A thermocouple was intentionally left in place at the prostate–SV junction to mark the internal treatment margin. Robotic bilateral seminal vesiculectomy with vas deferens excision was then performed en bloc, using the retained thermocouple to guide the proximal resection margin. He was discharged on postoperative day 1 after Foley removal.
Pathology and Follow-Up
Final pathology demonstrated bilateral SV and vas deferens involvement by Grade Group 3 (Pattern 4 = 80 percent) adenocarcinoma, with perineural and lymphovascular invasion. The right SV showed 70 percent involvement and the left SV 30 percent, confirming occult bilateral disease despite unilateral imaging findings. No radiation treatment effect was identified.
At 12 months, PSA remains stable at 0.10 ng/mL with no radiographic evidence of recurrence. He reports stable urgency/frequency without urinary incontinence.
Discussion
Prostate Cancer in 2026: The Scale of the Problem. Prostate cancer remains the most commonly diagnosed non-cutaneous malignancy among men in the United States. For 2026, the American Cancer Society estimates 333,830 new cases and 36,320 deaths, and reports that prostate cancer incidence has risen by about 3 percent annually since 2014 after a prior decline.1
The majority of these new diagnoses are detected at a localized or regional stage, where curative-intent local therapies—radical prostatectomy and radiation therapy—remain the cornerstones of treatment.
Radiation therapy, delivered as external beam radiation therapy (EBRT), stereotactic body radiation therapy (SBRT), brachytherapy, or in combination, is used as primary curative-intent treatment in approximately one-third of men with localized prostate cancer. Applying this proportion to 2026 incidence projections, more than 100,000 men in the United States are expected to undergo primary radiation therapy this year alone.
Although contemporary radiation therapy provides durable cancer control for most men, biochemical recurrence remains clinically relevant, particularly among men with intermediate- and high-risk features (approximately 10 to 15 percent in intermediate-risk disease at seven years in contemporary SBRT series, with higher rates in men with adverse baseline features).2,3 Extrapolating these rates, tens of thousands of men annually may ultimately face radiorecurrent disease, with a subset harboring biopsy-proven local recurrence potentially amenable to salvage intervention.
From Localization to Salvage: The Role of mpMRI. Historically, localization of recurrent prostate cancer after primary radiation therapy was imprecise, and salvage options were often limited to androgen deprivation therapy or salvage radical prostatectomy. Although salvage radical prostatectomy can provide durable cancer control in carefully selected patients, it has traditionally been associated with substantial morbidity, particularly urinary incontinence, anastomotic stricture, and, less commonly, rectal injury or fistula. These risks contributed to the underuse of curative-intent local salvage in men with radiorecurrent disease.4,5
Advances in mpMRI, PSMA PET imaging, and targeted biopsy have fundamentally changed the evaluation of radiorecurrent disease. Much as mpMRI transformed the diagnostic pathway for newly diagnosed prostate cancer, these technologies now allow more precise localization of recurrent tumor, improved assessment of disease extent, and better selection of patients for local salvage therapy.
This improved disease localization has expanded the role of salvage focal therapies, including cryoablation, high-intensity focused ultrasound (HIFU), brachytherapy, and reirradiation. In the MASTER meta-analysis by Valle et al. salvage local therapies demonstrated broadly similar five-year biochemical recurrence-free survival, with important differences in toxicity profiles. Salvage cryotherapy provided cancer control comparable to other accepted local salvage options while offering a less morbid alternative to salvage surgery in appropriately selected patients.6,7,8
The Challenge of Seminal Vesicle Recurrence. SV recurrence represents a particularly challenging pattern of radiorecurrent prostate cancer. In a detailed mapping study of 77 salvage radical prostatectomy specimens, Takeda et al. found SV involvement in 42 percent of cases, and most SV-positive patients also had concurrent intraprostatic disease. Their findings also highlighted frequent recurrence in the distal apex and periurethral region—areas not routinely sampled on standard biopsy—underscoring the multifocal nature of radiorecurrent disease and the importance of comprehensive evaluation, including targeted SV assessment when clinically suspected.9
SV recurrence is also technically difficult to treat. Salvage ablation alone may be limited by the risk of thermal injury to adjacent structures, including the ureter and posterior bladder wall, while surgical excision alone may leave residual disease at the prostate–SV junction. In a series of robotic salvage seminal vesiculectomy for isolated SV recurrence after brachytherapy, Langley et al. reported a 41 percent positive margin rate and 53 percent biochemical disease-free survival at three years, highlighting the limitations of surgery alone in this setting.10 Together, these data support the need for treatment strategies that address both the SV and the adjacent prostate base.
The NYU Langone Combined Approach: Rationale and Early Outcomes. To address this clinical challenge, our group at NYU Langone developed a combined salvage approach incorporating focal cryoablation and robotic seminal vesiculectomy. As first reported by Smigelski et al. in 2023, this technique is designed to treat both components of SV-associated recurrence: cryoablation targets residual intraprostatic disease and the prostate–SV junction, while robotic excision provides definitive removal of involved SV tissue. A key technical feature is retention of a thermocouple at the cryoablation margin, which serves as an intraoperative landmark to guide the proximal extent of robotic dissection and ensure continuity between the ablative and surgical treatment zones.11
In the initial NYU Langone series of seven patients, the combined approach was safe and well tolerated. There were no surgical complications, all patients were discharged on postoperative day one, and no patient developed new urinary incontinence. Among men with preserved baseline erectile function, potency was maintained. At a median follow-up of 1.4 years, 71 percent of patients remained disease-free on PSA and mpMRI surveillance. Notably, among the patients with recurrence, several had isolated contralateral SV recurrence that was successfully managed with repeat salvage treatment, highlighting both the feasibility of this approach and the complexity of SV-associated radiorecurrent disease.11
Lessons from the Current Case. This case reinforces and extends several key observations from the initial NYU Langone series. Most notably, despite preoperative mpMRI, PSMA PET, and targeted biopsy suggesting recurrence confined to the right SV, final pathology demonstrated bilateral SV and vas deferens involvement with Grade Group 3 disease, extensive pattern 4, perineural invasion, and lymphovascular invasion. This finding of occult contralateral disease is consistent with the Takeda mapping data and with the pattern of contralateral SV recurrence observed in three of four recurrent cases in the Smigelski series.9,11 This highlights an important limitation of current imaging and biopsy strategies: even with contemporary imaging, microscopic contralateral SV involvement may go undetected.
This observation also has an important practical implication. In selected patients undergoing combined salvage focal cryoablation and seminal vesiculectomy for SV-associated recurrence, bilateral vesiculectomy should be performed even when imaging suggests unilateral disease. At the same time, this recommendation should be viewed as hypothesis-generating rather than definitive, given the limited available experience.
The case also demonstrates the feasibility and tolerability of this combined approach in a heavily pre-treated patient—one who had undergone IMRT, salvage pelvic nodal radiation, HDR brachytherapy, and a period of ADT. The procedure was well tolerated, required only a one-day hospitalization, and has produced a stable PSA of 0.10 ng/mL through 12 months of follow-up. The absence of radiation treatment effect in the excised SV specimens—despite multiple prior radiation exposures—further highlights the capacity of the SVs to harbor radioresistant disease and the value of surgical excision as a component of the salvage strategy.
Evolving Landscape and Future Directions. The 2024 AUA/ASTRO/SUO Salvage Therapy Guideline reflects the growing role of advanced imaging and individualized treatment planning in the management of radiorecurrent prostate cancer.12 As mpMRI, PSMA PET, and targeted biopsy continue to improve localization of recurrent disease, more men with isolated local recurrence are likely to be identified as candidates for curative-intent salvage intervention.
The combined focal cryoablation and robotic seminal vesiculectomy approach represents an innovative extension of the modern salvage focal therapy paradigm for a particularly challenging pattern of recurrence. This case, together with the initial NYU Langone experience, supports its feasibility as a minimally invasive option in carefully selected patients with SV involvement.
Further prospective study with longer follow-up is needed to better define patient selection, optimize technical execution, and establish long-term oncologic outcomes. Until such data are available, the case presented here and the initial Smigelski series11 support the consideration of this combined technique as a viable and minimally invasive salvage option for appropriately selected men.
Conclusion
Salvage cryoablation combined with robotic seminal vesiculectomy offers a novel and promising approach for managing locally recurrent prostate cancer with SV involvement following primary radiation therapy. As imaging capabilities and awareness of SV recurrence expand, this combined technique warrants broader consideration in the management of radiorecurrent prostate cancer with SV involvement.
References
- Siegel RL, et al. CA Cancer J Clin. 2026. DOI.
- Kishan AU, et al. JAMA Netw Open. 2019;2(2):e188006. DOI.
- Gogineni E, et al. Int J Radiat Oncol Biol Phys. 2020;109:1232-1242. DOI.
- Sanderson KM, et al. J Urol. 2006;176(5):2025-2031. DOI.
- Chade DC, et al. Eur Urol. 2011;60(2):205-210. DOI.
- Valle LF, et al. Eur Urol. 2021;80(3):280-292. DOI.
- Chin JL, et al. J Urol. 2021;206(3):646-653. DOI.
- Tan WP, et al. Clin Genitourin Cancer. 2020;18(3):e260-e265. DOI.
- Takeda T, et al. Eur Urol. 2018;73(4):488-495. DOI.
- Langley S, et al. BJU Int. 2022;129(6):731-738. DOI.
- Smigelski MB, et al. J Endourol. 2023;37(8):876-881. DOI.
- Morgan TM, et al. J Urol. 2024;211(4):509-517. DOI.